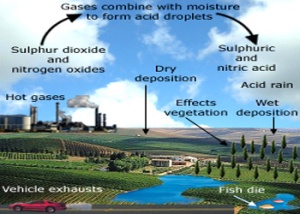
There are two major types of acid deposition: wet deposition and dry deposition. Wet-acid deposition is more commonly known as acid rain. The creation of wet-acid deposition starts when harmful gasses from factories and cars are emitted into the air. Specifically the chemicals that cause acid deposition are sulfur dioxide and nitrogen dioxide. These chemicals rise into the atmosphere where they interact with moisture. This converts the gasses into sulfuric acid and nitric acid. The acid then returns to the earth in the form of rain or snow. Dry-acid deposition occurs when gasses and particles attach to the surfaces of plants, soil, and water.(1)
Figure 1 (http://www.wwk.in/acid-rain-theme)
The effects of acid deposition are very detrimental to the environment. Acid deposition reduces the pH of water from 5.5-6 to below 5. This change in pH produces lethal conditions for many marine organisms. When a group of organisms in an ecosystem die, the whole ecosystem suffers. Even if it is a small, seemingly harmless organism, its absence can cause a trophic cascade. (1) Acid deposition is detrimental for plants and forests because it gets rid of essential nutrients in the soil such as magnesium and calcium. In the absence of these nutrients, the trees are more vulnerable to catch an infection and become damaged by weather or insects. Acid rain also wears on the outer layer of leaves. If leaves are damaged, the plant or tree cannot perform photosynthesis, which is the pathway that produces food and energy. Dry-acid deposition is also a threat for humans. The inhalation of airborne gasses and particles causes respiratory problems and diseases such as asthma, bronchitis, and phenomena.(2)
Needless to say, acid deposition is not good for the environment or for humans. To combat the amount of acid deposition occurring, emissions into the atmosphere need to be reduced. A large source of sulfur and nitrogen dioxide comes from the burning of coal in power plants. To reduce the problem of gasses being released into the atmosphere, many facilities have installed “coal scrubbers”. These filters pass the hot gas through a limestone mixture, which reacts with the acidic gasses and removes them before they leave the smokestack. (1)
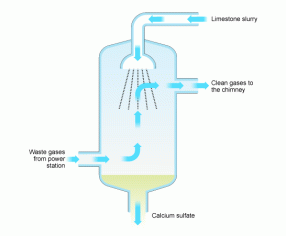
Figure 3 (http://www.bbc.co.uk/schools/gcsebitesize/science/aqa_pre_2011/rocks/fuelsrev5.shtml)
Another source of sulfuric acid that is not caused by human activity is volcanic eruptions. For example, when Mount Pinatubo in the Philippines erupted in 1991, 20 million tons of sulfur dioxide along with ash was blasted into the atmosphere. The blast pushed the gas and particles up to 12 miles high into the atmosphere. (3)
Figure 3 (http://earthobservatory.nasa.gov/Features/Volcano/)
Whether the emission of harmful gasses is natural or not, the creation of acid deposition is terrible for the environment. While we cannot control volcanic eruptions, we can control our emissions into the atmosphere by innovations such as the coal scrubber. More progress must be made so that there is no further damage to ecosystems around the world.


Hannah,
This is a very interesting and informative post. I was only aware of acid rain, or “wet deposition,” and I knew it harmed the environment somehow (based on the name), but I had no idea how or what a major impact it actually has. I also had no idea there was a second type of dry acid deposition, so I appreciate your explanation. I think it was important how you mentioned the affect acid rain has on the marine ecosystem, and explained how even organisms that may seem small and unimportant still have a major affect in the trophic system. Also, I was unaware of the effect acid deposition had on humans. This is just another example of how when humans impact or harm the environment, it can come back and harm us and many other organisms on Earth, I am glad that that we are beginning to try and reverse these affects of acid deposition with new inventions such as the coal scrubbers, and I hope with the technology and advances in science we have today, we can come up with more effective solutions to this serious problem!
Keep posting!
– Kristin
LikeLike